- This topic is empty.
-
AuthorPosts
-
03/04/2026 at 15:57 #5221
Medical device CDMO services is an outsourcing service model that provides full-process solutions from R&D to commercialization for the medical device industry. Its core is to help medical device enterprises reduce costs, shorten the research and development cycle, improve product quality, and accelerate the product market process through professional, large-scale and integrated services.
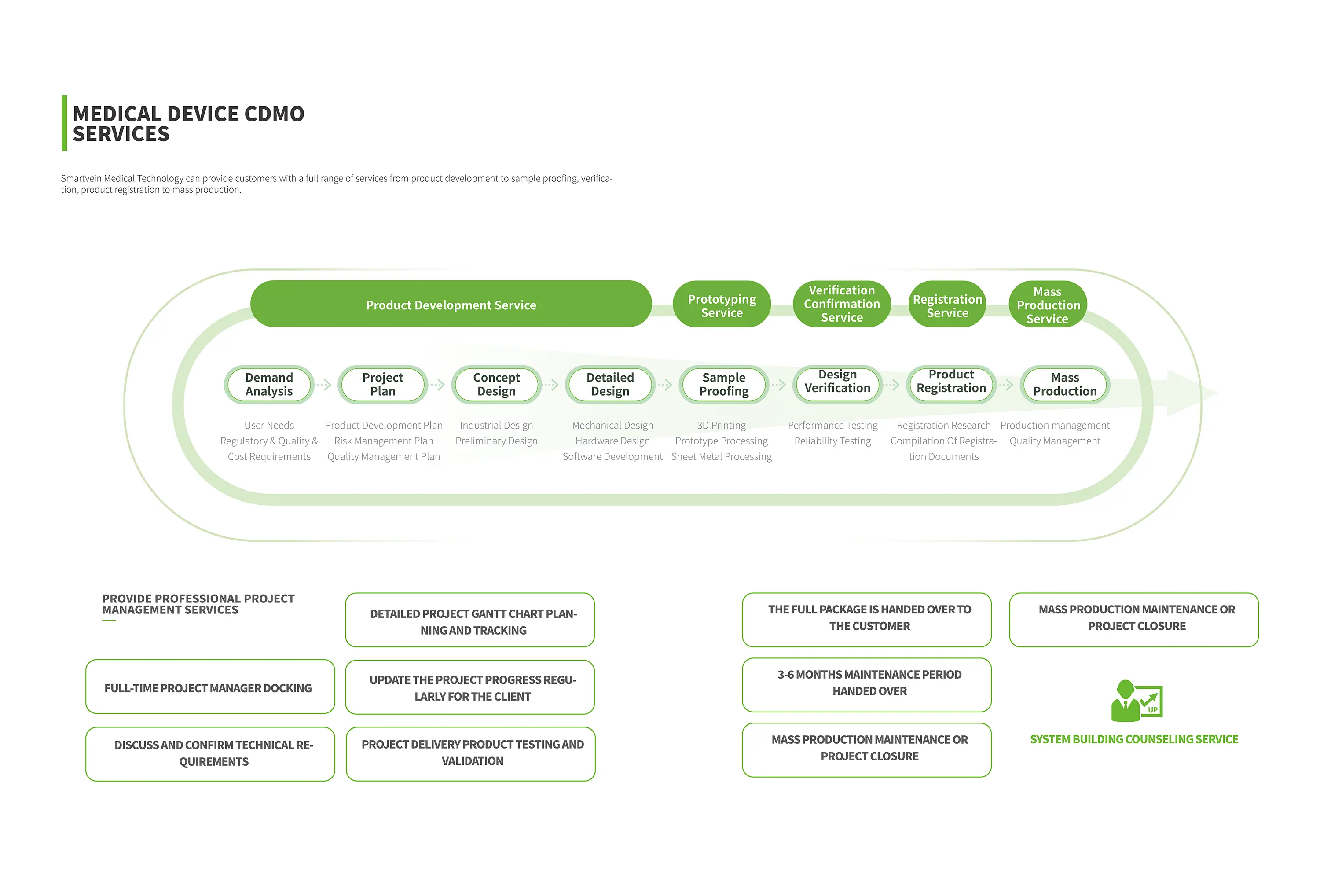
1. Definition and Characteristics of CDMO Services
Medical device CDMO is a new outsourcing service model, mainly for medical device enterprises to provide R & D design, process development, manufacturing, registration and declaration, quality control and other full chain services. This model allows medical device companies to outsource some or all of their research and development and production tasks to specialized organizations, thereby focusing on the improvement of core competitiveness.
2. Service Content
The scope of medical device CDMO's services covers the entire product lifecycle, including but not limited to the following:
– R&D Sage: demand analysis, laboratory research, product design, process development, sample production, etc.
– Production Stage: mass production, quality control, supply chain management, warehousing and logistics, etc.
– Registration Stage: regulatory consultation, clinical trial, registration declaration, quality system establishment, etc.
– Commercialization Stage: marketing, sales support and follow-up management.
3. Strengths and Values
– Cost-Effectiveness: By outsourcing non-core business, medical device companies can reduce equipment investment and production line construction, saving research and development and production costs.
– Technical Advantages: CDMO enterprises usually have advanced technology and equipment, and can provide high-quality products and services.
– Flexibility and Efficiency: CDMO model has high flexibility, can quickly adjust the service content according to customer needs, while shortening the product development cycle.
– Risk Control: reduce technical risks and financial pressure in R&D and production through specialized division of labor.
4. Industry Background and Development Trend
With the full implementation of the medical device Registrant system (MAH), as well as the promotion of market demand and technological progress, the medical device CDMO industry has ushered in a period of rapid development. More and more enterprises choose CDMO services to accelerate the process of product market and enhance competitiveness.
5. Typical Application Scenarios
– Start-Ups: Companies that lack R&D and production capabilities can quickly develop and go to market with CDMO services.
– Mature Companies: Companies that want to optimize production processes and reduce costs can increase their competitiveness with CDMO services.
– Localization of Imported Products: Through the CDMO platform, imported medical devices can achieve localized production and shorten the market cycle.
6. Future Outlook
The medical device CDMO industry is still in the growth stage, and is expected to further expand its scale through technological innovation and industrial chain integration in the future. With the increase of policy support and market demand, this field will attract more enterprises to participate and promote the high-quality development of the entire industry.
Medical device CDMO services is an efficient, flexible and widely used outsourcing model. It can not only help medical device enterprises optimize resource allocation, reduce research and development costs, but also accelerate the transformation process of products from research and development to the market, so as to occupy a favorable position in the fierce market competition.
http://www.smartveingroups.net
SmartVein Group -
AuthorPosts
- You must be logged in to reply to this topic.